FDA allows Houston cancer doctor to resume drug trial
Por um escritor misterioso
Descrição
Federal regulators have lifted a partial hold on a clinical trial performed by Stanislaw

CV Disease Fellowship: Fellows

Promising Medical Advances in Cancer Treatment and Care

Katy Rezvani MD Anderson Cancer Center

Lurbinectedin as second-line treatment for patients with small-cell lung cancer: a single-arm, open-label, phase 2 basket trial - The Lancet Oncology
FDA clears MD Anderson to test skin cancer treatment on humans

Study finds why some cancer drugs may be ineffective - UTHealth News - UTHealth Houston

Hagop M. Kantarjian MD Anderson Cancer Center

World's First Gene Therapy Clinical Trial for Glycogen Storage Disease Approved by FDA - Connecticut Children's

When Cancer Patients Ask About Weed, Many Doctors Say Go For It – Houston Public Media

Doctor claims to cure pediatric cancer, critics skeptical

PHL Inno - Lab Notes: Larimar plans $70M stock sale, gets FDA clearance to resume clinical testing
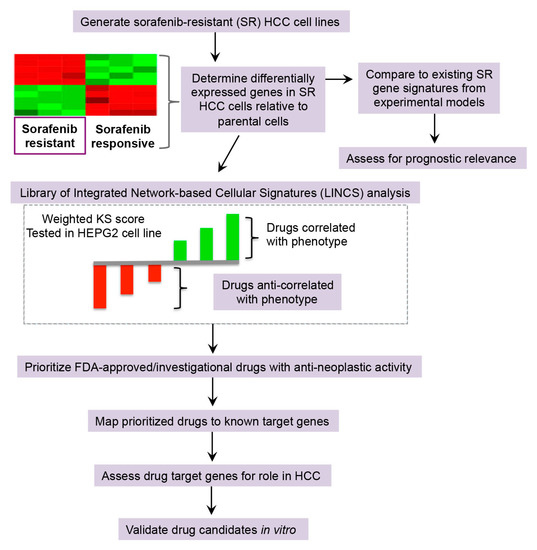
Cancers, Free Full-Text

Texas judges clear Houston cancer doctor of most medical misconduct charges
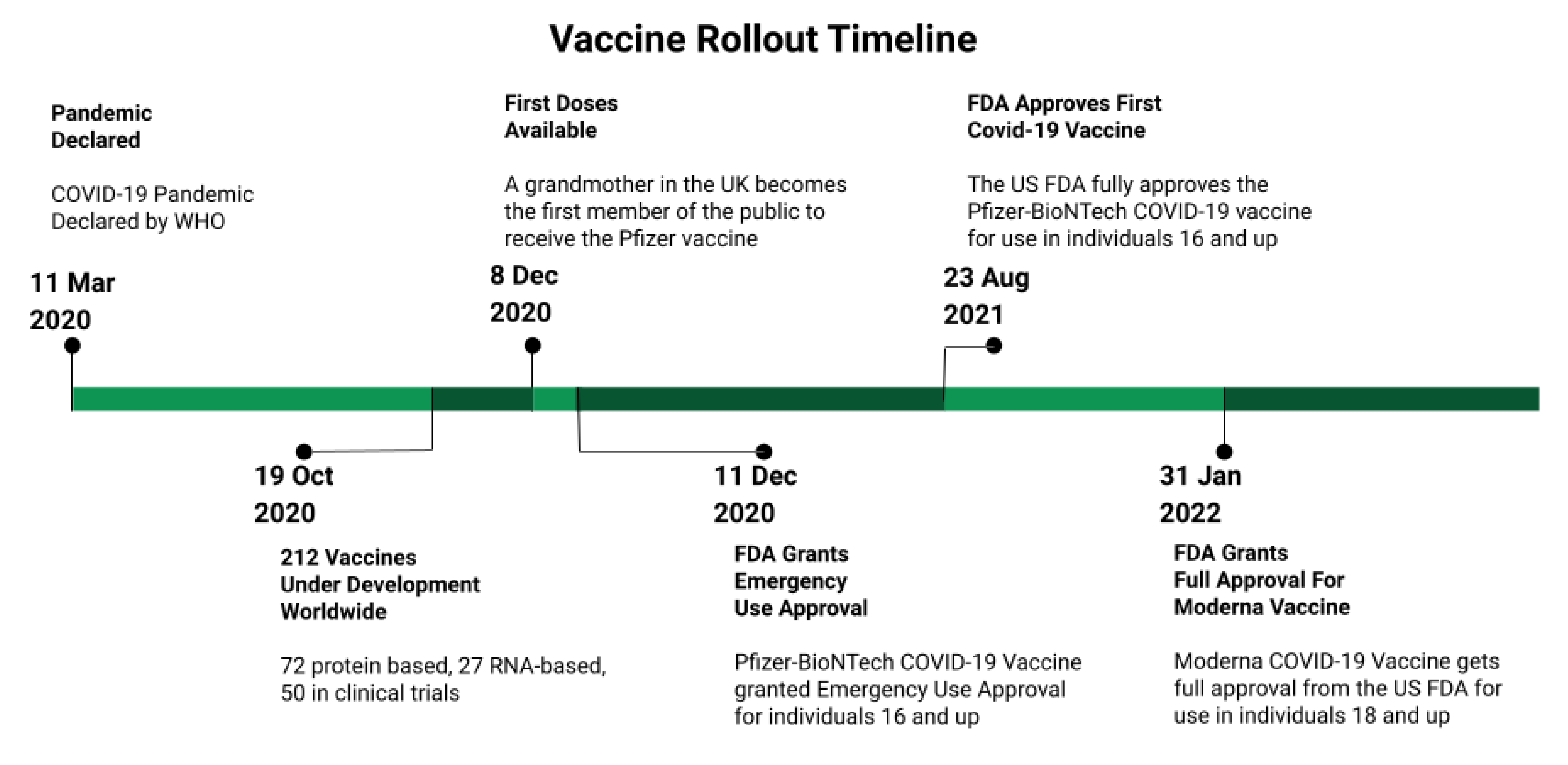
Vaccines, Free Full-Text

FibroGen limited access to Duchenne muscular dystrophy drug after failed clinical trial. But mom says it works for her son. - San Francisco Business Times
de
por adulto (o preço varia de acordo com o tamanho do grupo)







